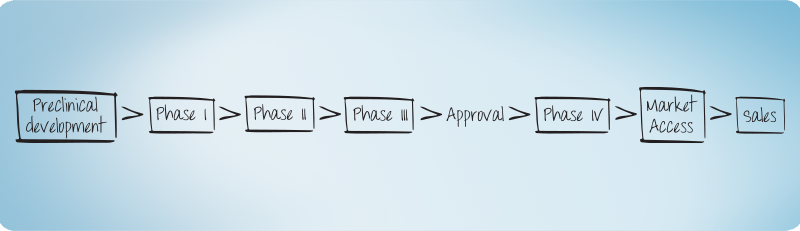
Clinical Research
Enjoy the difference of having a proactive partner.
From proposal, through the project all the way to final reflections a dedicated team will take care of your project's tasks and utilize relevant specialists.
AMS Clinical Research consists of Project Managers, Clinical Research Associates, Regulatory specialists, Physicians, Statisticians, Datamanagers, a centralized Project Administration and a dedicated Quality and Development group. All of AMS staff brings a wealth of experience and high commitment to your service.
AMS Clinical Research combines state-of-the-art high quality processes with innovative technologies. Our clients benefit from reliable project delivery, robust data and modern project designs.
Read more or find out how AMS Clinical Research services will meet your needs: