Clinical Research - read more
Enjoy the difference of having a proactive partner.
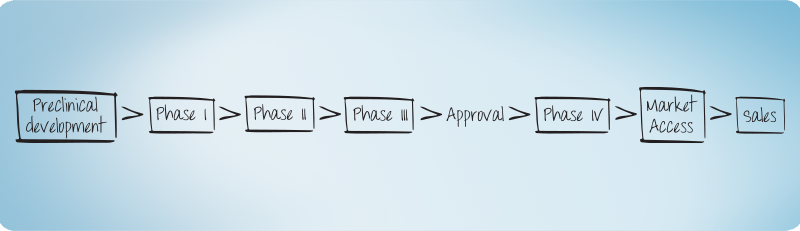
AMS provides full Clinical Research services including post-marketing services with a focus in Europe and global reach. For global coverage, for long term AMS collaborates with organizations to provide all the resources and working structures needed for your project.
Whatever the task, our clients have total confidence in AMS Clinical Research:
- excellence in planning and implementation to deliver solutions more quickly
- reliability and commitment to timelines, quality and budget
- compliance with the latest regulatory requirements
- efficiency in study set-up, data management including Electronic Data Capture (EDC), and analysis
The experience, solution-oriented and flexible thinking, and proactivity of AMS teams will stand out from the crowd from the moment the team is presented to you through to the final report.